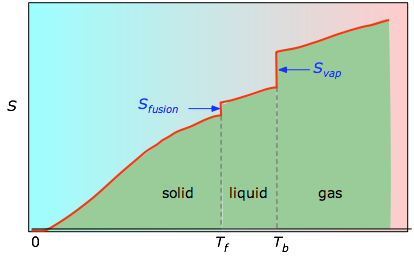
When latent heat is added, no temperature change occurs. This energy breaks down the intermolecular attractive forces, and also must provide the energy necessary to expand the gas (the pΔV work). Latent heat is the amount of heat added to or removed from a substance to produce a change in phase. In case of liquid to gas phase change, this amount of energy is known as the enthalpy of vaporization, (symbol ∆H vap unit: J) also known as the (latent) heat of vaporization or heat of evaporation. In general, when a material changes phase from solid to liquid, or from liquid to gas a certain amount of energy is involved in this change of phase. It vanishes completely at a certain point called the critical point. To produce of 1 kg of saturated steam from feedwater, about 1789/206 x 1 kg = 8.68 kg of primary coolant is required.Įnthalpy of Vaporization The heat of vaporization diminishes with increasing pressure, while the boiling point increases. Since the difference in specific enthalpies is less for primary coolant than for feedwater, it is obvious that the amount of primary coolant will be higher than 1kg. Feedwater is then evaporated and the pressurized steam ( saturated steam 280☌ 536☏ 6,5 MPa) leaves the steam generator through steam outlet and continues to the steam turbine. 
The feedwater (secondary circuit) is heated from ~230☌ 446☏ to the boiling point of that fluid (280☌ 536☏ 6,5MPa). The feedwater ( water 230☌ 446☏ 6,5MPa) is pumped into the steam generator through the feedwater inlet. The primary coolant leaves (water 295☌ 563☏ 16MPa) the steam generator through primary outlet. 
The hot primary coolant ( water 330☌ 626☏ 16MPa) is pumped into the steam generator through primary inlet. Assume that there are no energy losses, this is only idealized example. The fact that the absolute value of specific enthalpy is unknown is not a problem, however, because it is the change in specific enthalpy (∆h) and not the absolute value that is important in practical problems.Ĭalculate the amount of primary coolant, which is required to evaporate 1 kg of feedwater in a typical steam generator. For example, the specific enthalpy of water or steam is given using the reference that the specific enthalpy of water is zero at 0.01☌ and normal atmospheric pressure, where h L = 0.00 kJ/kg. Normally, the enthalpy of a substance is given with respect to some reference value. In general, enthalpy is a property of a substance, like pressure, temperature, and volume, but it cannot be measured directly. The specific enthalpy is equal to the specific internal energy of the system plus the product of pressure and specific volume. Note that the enthalpy is the thermodynamic quantity equivalent to the total heat content of a system. It equals to the total enthalpy (H) divided by the total mass (m). The specific enthalpy (h) of a substance is its enthalpy per unit mass. Engineers use the specific enthalpy in thermodynamic analysis more than the enthalpy itself. The enthalpy can be made into an intensive, or specific, variable by dividing by the mass. Vaporization (or Evaporation) the transition of molecules from a liquid to a gaseous state the molecules on a surface are usually the first to undergo a phase change.Enthalpy in Intensive Units – Specific Enthalpy Extensive and intensive properties of medium in the pressurizer. The list of enthalpies of vaporization given in the Table T5 bears this out. We can thus expect liquids with strong intermolecular forces to have larger enthalpies of vaporization. In general the energy needed differs from one liquid to another depending on the magnitude of the intermolecular forces. Such a separation requires energy (in the form of heat). Heat is absorbed when a liquid boils because molecules which are held together by intermolecular attractive interactions and are jostled free of each other as the gas is formed. \) is the enthalpy of the liquid state of a compound or element
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
